Data Management & Cloud-based QMS Go Hand-in-Hand

Data and data management have been the backbone of business since its inception. The first Neanderthal that successfully opened a meat roasting business figured out, though they may not have understood it, that data was the key to their success. They had to have the right cave, in the right place, with the right size fire, cooking the right amount of mammoth meat. Data! Ok, that might be going a bit too far back, but the principle holds true.
Data management and data integrity are the kings of all good business decisions.
In the Life Sciences industry, that concept compounds exponentially. Not only do you have to deal with business data but you also have to keep track of regulatory and scientific data. Which is where a quality QMS comes into play.
As stated by the PDA, “a key cornerstone for the integration of Data Integrity into a robust Quality Management System (QMS) capable of preventing, detecting, and remediating Data Integrity issues will require keeping a pulse on Regulatory guidance and trends to ensure both; knowledge of the requirements, as well as how the requirements continue to be interpreted.” In legacy QMS systems as well as paper-based solutions, managing all that can easily become a herculean task. It requires an entire team of people just to collect, analyze, and maintain it. The costs can quickly become astronomical, and the ability to provide actionable data of effective quality controls becomes unattainable . Especially if any type of change happens in your business model. Data can easily become lost through the cracks of updating your legacy system.
That is where a cloud-based QMS makes a huge difference.
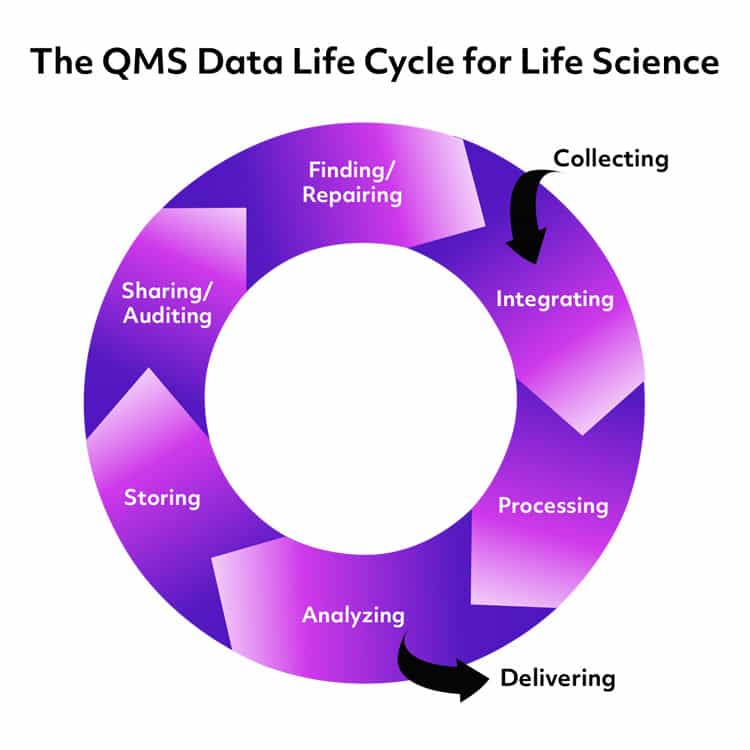
Data is what QMS is built around. And, data is one of the most basic building blocks of a ready-to-use QMS. Collecting it. Managing it. Protecting it. As well as initiating corrective actions based on it. It understands that your business can’t reach its full potential without well managed and protected data. Here are the main ways a cloud based QMS deals with your data.
1. Data Collection –
All businesses have a lot of data but Life Science businesses have ridiculous amounts of it. And, it’s not all the same kind of data. A ready-to-use QMS handles that very important and simple fact. It has been designed to allow you to collect data, whether it’s about mitigating risks for ISO 9001 or regulatory commitment tracking. The QMS gathers from a wide range of sources, including CAPA, deviations, suppliers, and product lifecycle monitoring.
2. Data Maintenance –
Getting data is only step one. The second is knowing that the data is maintained properly. Can your QMS store the data in a way that is easily accessible? Easy to validate? And of course, secure enough to avoid corruption? All of this while giving anyone who needs access access from almost anywhere?
3. Data Validation –
With the ability to give anyone in your company access to information, validation of data is a much simpler process. A cloud-based QMS allows you to grant access to any of the data it holds to however many people necessary to keep things moving. The system tracks users’ activity, so there will always be a trail of interactions.
4. Data Presentation –
Finally, there is no need to worry about presenting your data to any third parties that might need it. Whether it is via an audit or just for an office based presentation, you will be able to display your data in multiple ways that others can easily understand.
Data, in all its glory, is more manageable and presentable with a quality QMS.
All-in-all, you should not have any concern about how you handle data. Modern ready-to-use QMS specifically handles data in all the ways companies have dreamed of. No longer is it built as if it were from the stone age where things were tracked with cave paintings. With your system running on the cloud your QMS can steer your company into the future.
If you want to learn more about how a Ready-To-Use QMS can dramatically shape and improve the speed of your business, download our most recent eBook, The Need for Speed. It takes a deeper dive into how a Ready-To-Use QMS differs from a standard legacy or paper-based solution.