The Dot Compliance Blog
Your source for AI, quality, and compliance updates, insights and best practices across the life sciences industry.
All
21 CFR Part 11AnnouncementArtificial IntelligenceAudit ManagementBest PracticesBiotechnologyCAPAChange ManagementClinicalComplaint ManagementCosmeticsData and AnalyticsDocument ManagementeQMSISO 13485Life SciencesManufacturingMedical DevicePharmaceuticalQuality 4.0Regulatory ComplianceRisk ManagementSaaSTraining Management

How Is an eQMS Used in Biotech vs. Med Devices?
August 2025

Which Features Matter Most When Choosing an eQMS?
August 2025

How Is AI Changing the Way Life Sciences Companies Manage Quality?
July 2025
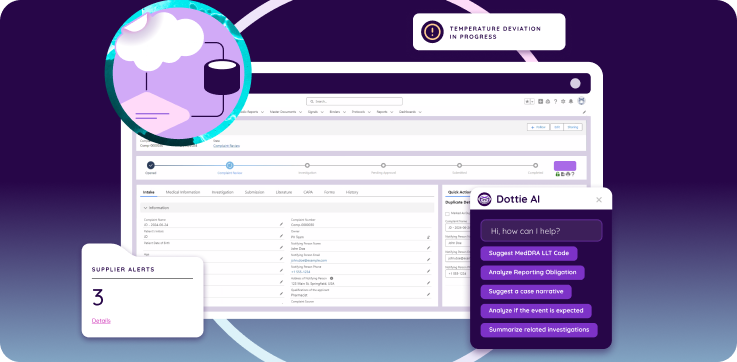
How Should a QMS Be Configured for GxP Compliance?
July 2025

Can a QMS Help Us Prepare for an FDA Inspection?
July 2025

What Documentation Does a QMS Need for FDA 21 CFR Part 11?
July 2025

Can a QMS Help You Pass an FDA Audit?
July 2025

Why Is an eQMS Critical for Life Sciences Companies in 2025?
July 2025

Can a QMS Automate CAPA Management?
July 2025

Why Do Medical Device Companies Need an ISO 13485-Compliant QMS?
July 2025

What QMS Features Should We Prioritize in 2025?
July 2025

What Does ISO 13485 Require for a Medical Device QMS?
July 2025