
“Dot Compliance has helped us be more efficient in our processes, while also maintaining a high level of compliance in a highly regulated industry.”



Life sciences organizations are reaping the benefits of electronic batch records to improve efficiency, ensure accuracy, and streamline operations.
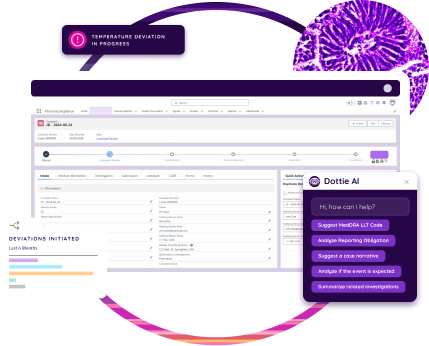
Electronic batch records seamlessly connect with systems like eQMS, LIMS, and ERP to create a unified digital environment. This integration eliminates data silos, streamlines workflows, and improves coordination across production, quality, and supply chain operations. By connecting critical systems, teams gain real-time visibility and control over every stage of manufacturing.
Electronic systems adapt easily to growing production volumes and evolving process complexity. Dot Compliance supports scalability by enabling organizations to add new products, sites, or workflows without disrupting existing operations. This flexibility helps companies stay agile and maintain efficiency as they expand.

Automated data entry and recordkeeping significantly reduce human error and ensure accuracy across all production records. Built-in validation rules verify inputs at every step, maintaining data integrity and regulatory compliance. With consistent, error-free documentation, teams can trust their records and accelerate batch release.


“Dot Compliance has helped us be more efficient in our processes, while also maintaining a high level of compliance in a highly regulated industry.”
Used by life sciences organizations around the world, Dot Compliance empowers teams to manage quality with confidence, maintain continuous compliance, and keep every process inspection ready.